Nilotinib Hydrochloride API
| CAS No. | 923288-90-8 |
| Therapeutic Category | Anti-Cancer/Oncology |
| Technology | Synthetic |
| Dosage Form | Oral Solids |
| Innovator Brand | Tasigna(USA) |
| Registration Status | Technical Package |
| Polymorph | Form B monohydrate; Form A dihydrate |
| GMP | FDA(USA)/NMPA(China)/MFDS(Korea) approved |
| EHS | ISO 14001 & 45001 certified |
Product Description
- Grade: Active Pharmaceutical Ingredient(API)
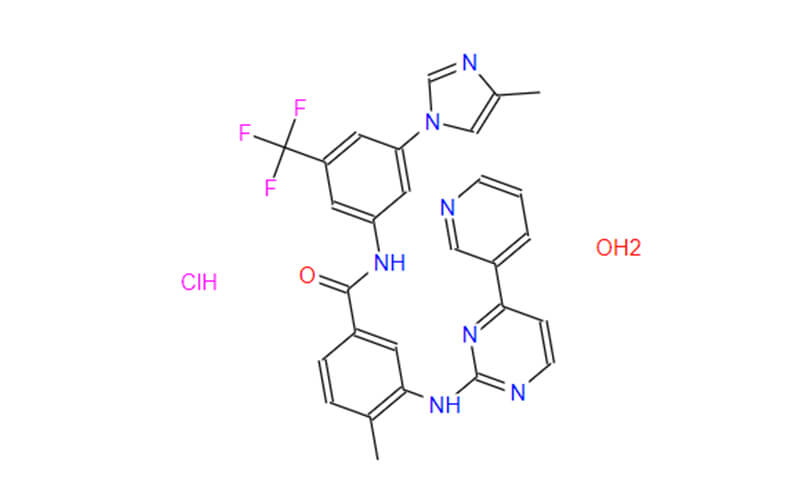
- Chemical Name: 4-methyl-N-[3-(4-methylimidazol-1-yl)-5-(trifluoromethyl)phenyl]-3-[(4-pyridin-3-ylpyrimidin-2-yl)amino]benzamide,hydrate,hydrochloride
- Molecular Formula: C28H25ClF3N7O2
- Molecular Weight: 583.992
- Specification: Enterprise Standard established according to ChP/USP/EP
- Appearance: Powder
- Total impurities: not more than 0.5%
- Purity: not less than 99%
- Residual Solvents: fully comply with ICH Q3C
- Mutagenic impurities: fully comply with ICH M8
- Nitrosamine assessment: available
- Particle size: regular grade or milling/sieving according to customer’s requirement.
- Storage: Room temperature
- Production capacity: Commercial
- Standard Package: 1kg/bag, 5kg/bag, or according to the customer’s requirement
Applications of Nilotinib API
- Nilotinib Hydrochloride Monohydrate is the monohydrate monohydrochloride form of nilotinib, an orally bioavailable aminopyrimidine-derivative Bcr-Abl tyrosine kinase inhibitor with antineoplastic activity. Designed to overcome imatinib resistance resulting from Bcr-Abl kinase mutations, upon administration, nilotinib binds to and stabilizes the inactive conformation of the kinase domain of the Abl portion of the Bcr-Abl fusion protein, resulting in the inhibition of the constitutive kinase activity of Bcr-Abl protein. This inhibits the Bcr-Abl-mediated proliferation of Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) cells. Nilotinib Hydrochloride also inhibits the receptor tyrosine kinases platelet-derived growth factor receptor (PDGF-R; PDGFR), mast/stem cell growth factor receptor Kit (c-Kit), and, to a lesser extent, colony-stimulating factor 1 receptor (CSF-1R; CSF1R), and discoidin domain-containing receptor 1 (DDR1).
Why Choose Us as Your Nilotinib Supplier?
- Qigmu is a Nilotinib supplier in China. Our factory is established according to ICH/USFDA/EU/JAPAN/China regulations and current GMP, totally complies with environmental law in China, no risk to removing the factory. A new API factory is under design and is predicted to be put into use in 2025.
- Qingmu’s team has rich experience in patent challenges on crystalline form & synthesis processes and also synthetic route development and scale-up & quality research.
- Qingmu’s lab is equipped with HPLC, GC, ICP-MS, GC-MS, CAD, microbalance, Malvern particle analyzer, etc. Qingmu passed the site inspection from NMPA(China), FDA(USA), and MFDS(Korea) and also customer audits from Europe, USA, Japan, etc.
- As a reliable Nilotinib API manufacturer, Qingmu successfully exported products to more than 40 countries including Japan, USA, Germany, Spain, the Netherlands, Russia, South Korea, and Brazil.